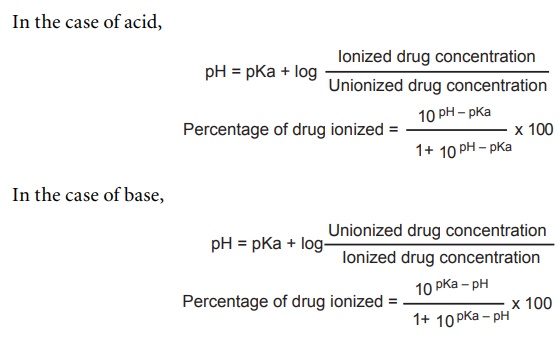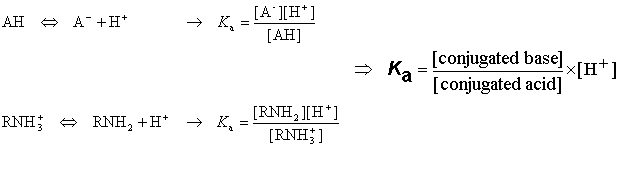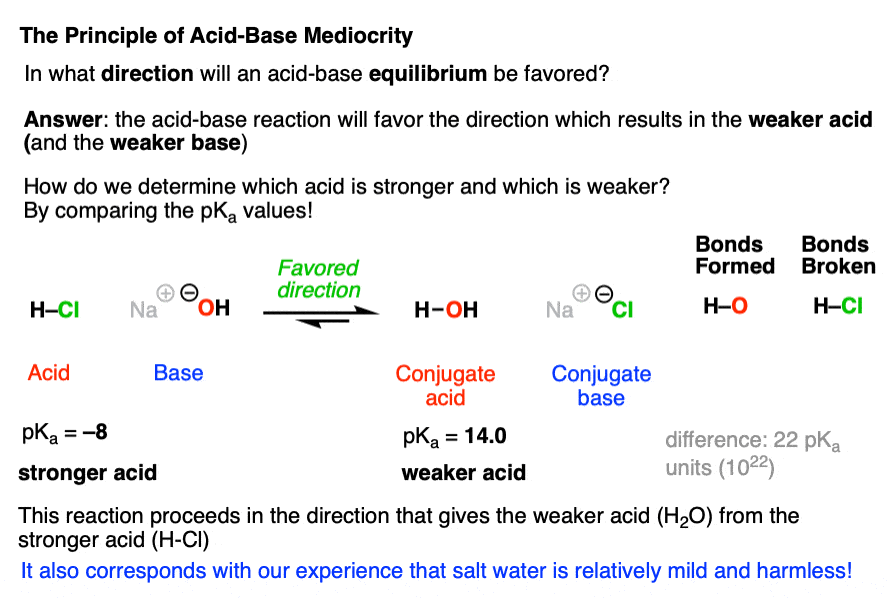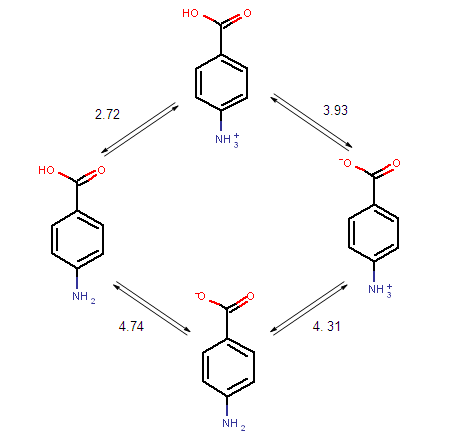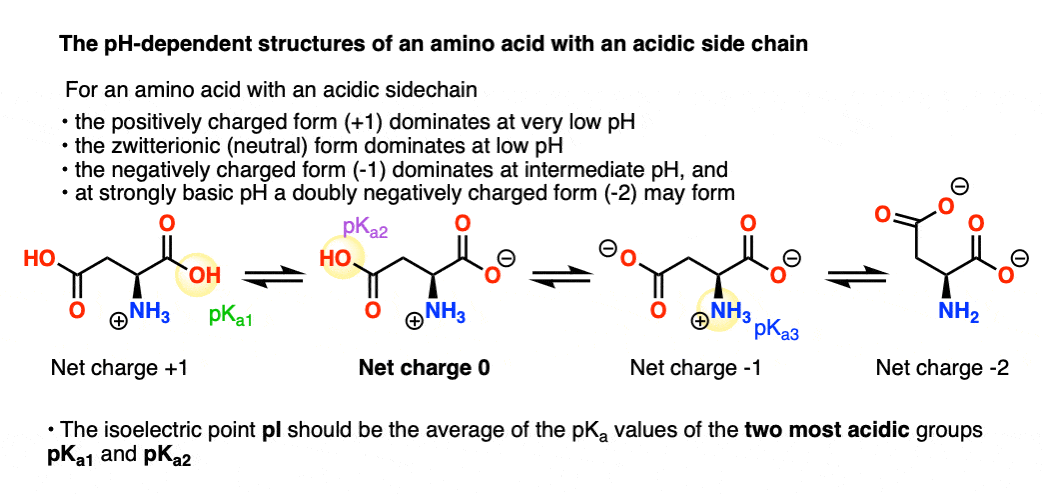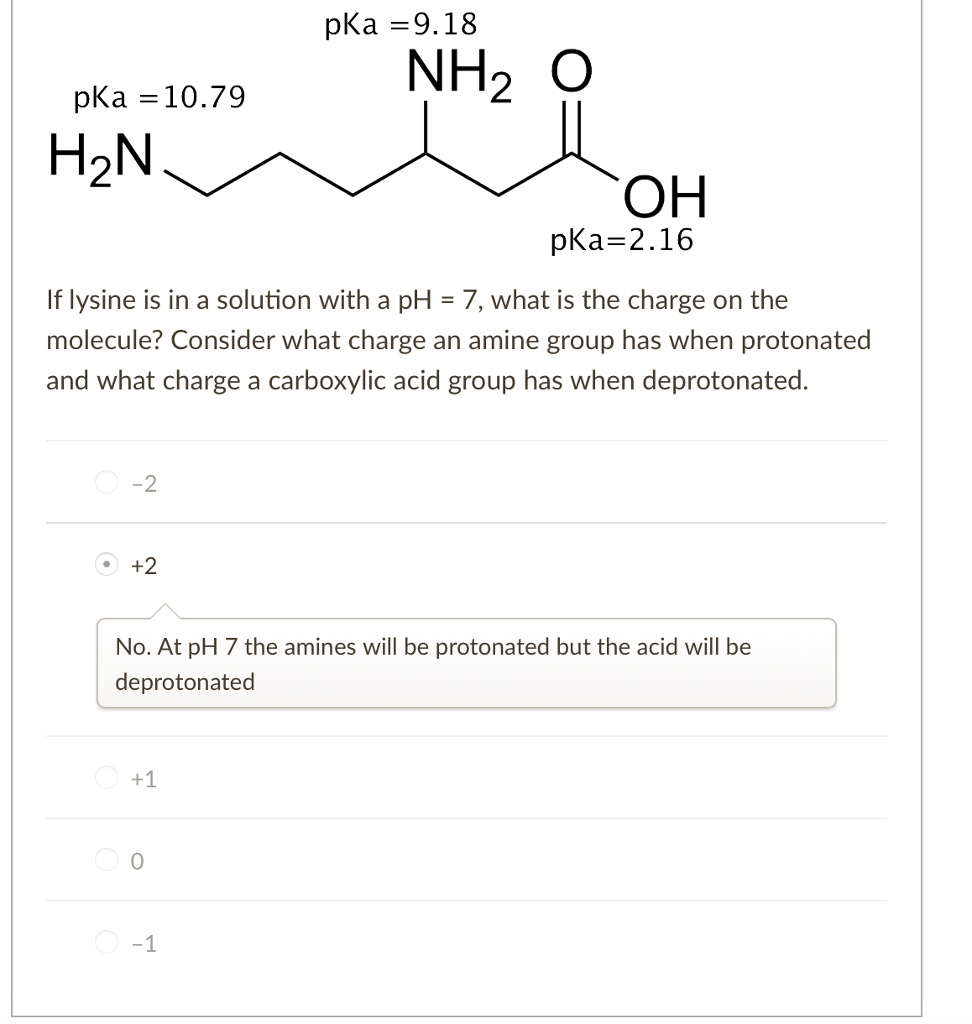
SOLVED: pKa =9.18 NH2 pKa =10.79 HzN OH pKa=2.16 If lysine is in a solution with a pH = 7, what is the charge on the molecule? Consider what charge an amine

A plot of the pH dependence of the solubility of an acid with pKa 4.8... | Download Scientific Diagram

First Principles pKa Calculations on Carboxylic Acids Using the SMD Solvation Model: Effect of Thermodynamic Cycle, Model Chemistry, and Explicit Solvent Molecules | The Journal of Physical Chemistry B
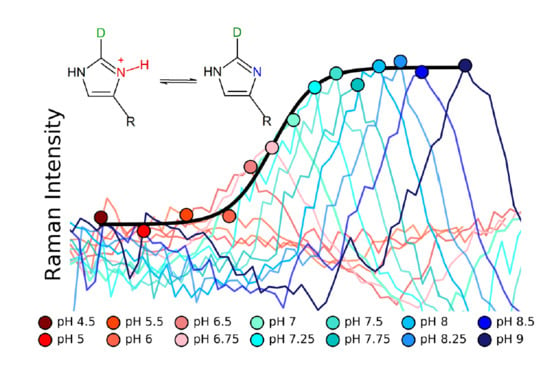
Molecules | Free Full-Text | pKa Determination of a Histidine Residue in a Short Peptide Using Raman Spectroscopy
![SOLVED: You willneed the following equations to help with this assignment: n = M n C = vol pH = 1ogi [H;O' ] pKa =10g10 Ka [A] pH = pKa 1og10 ` [ SOLVED: You willneed the following equations to help with this assignment: n = M n C = vol pH = 1ogi [H;O' ] pKa =10g10 Ka [A] pH = pKa 1og10 ` [](https://cdn.numerade.com/ask_images/da943d40c18e4feb99e9ecb5f7eb7cd6.jpg)
SOLVED: You willneed the following equations to help with this assignment: n = M n C = vol pH = 1ogi [H;O' ] pKa =10g10 Ka [A] pH = pKa 1og10 ` [

:max_bytes(150000):strip_icc()/GettyImages-536839741-56a135503df78cf7726864c3.jpg)