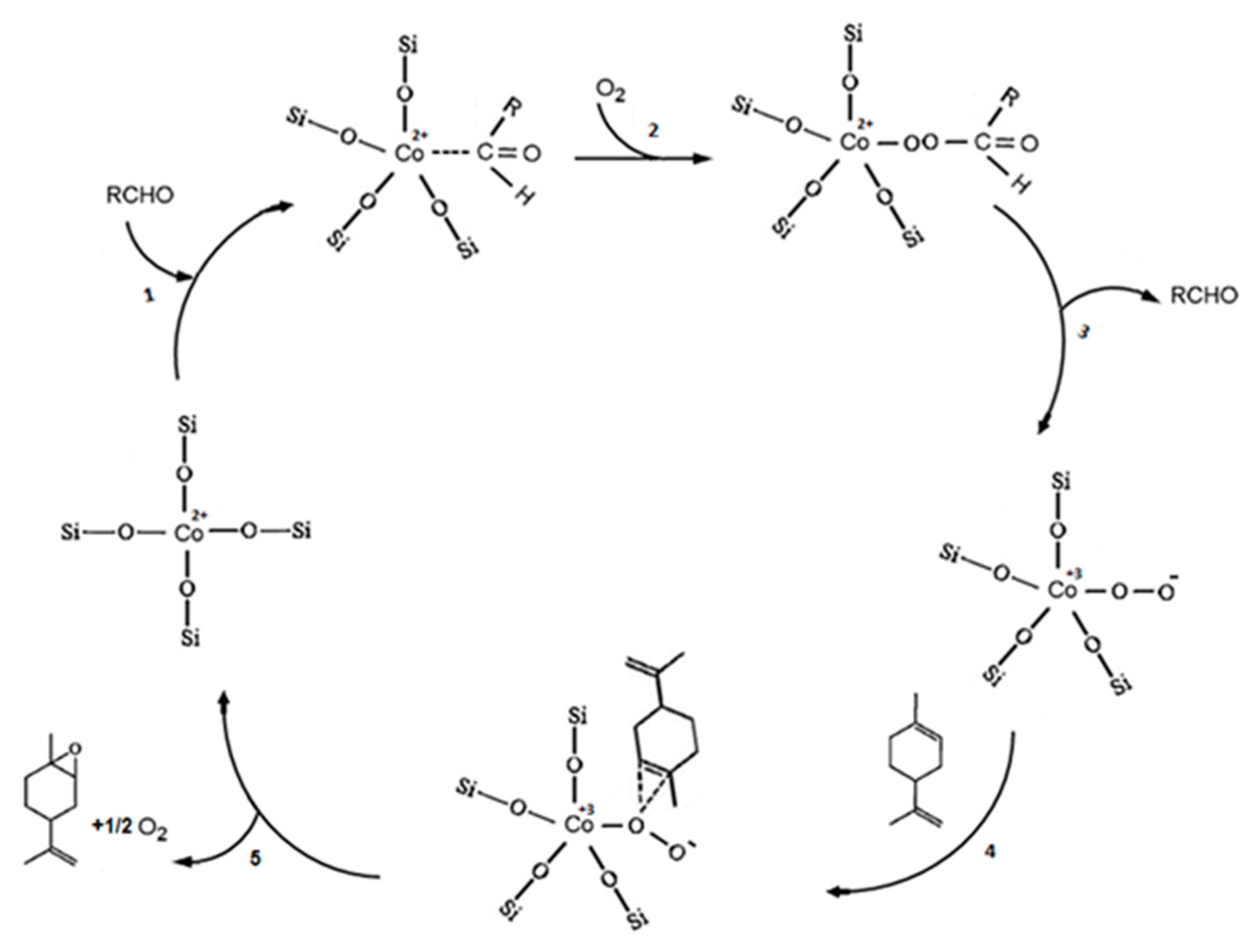
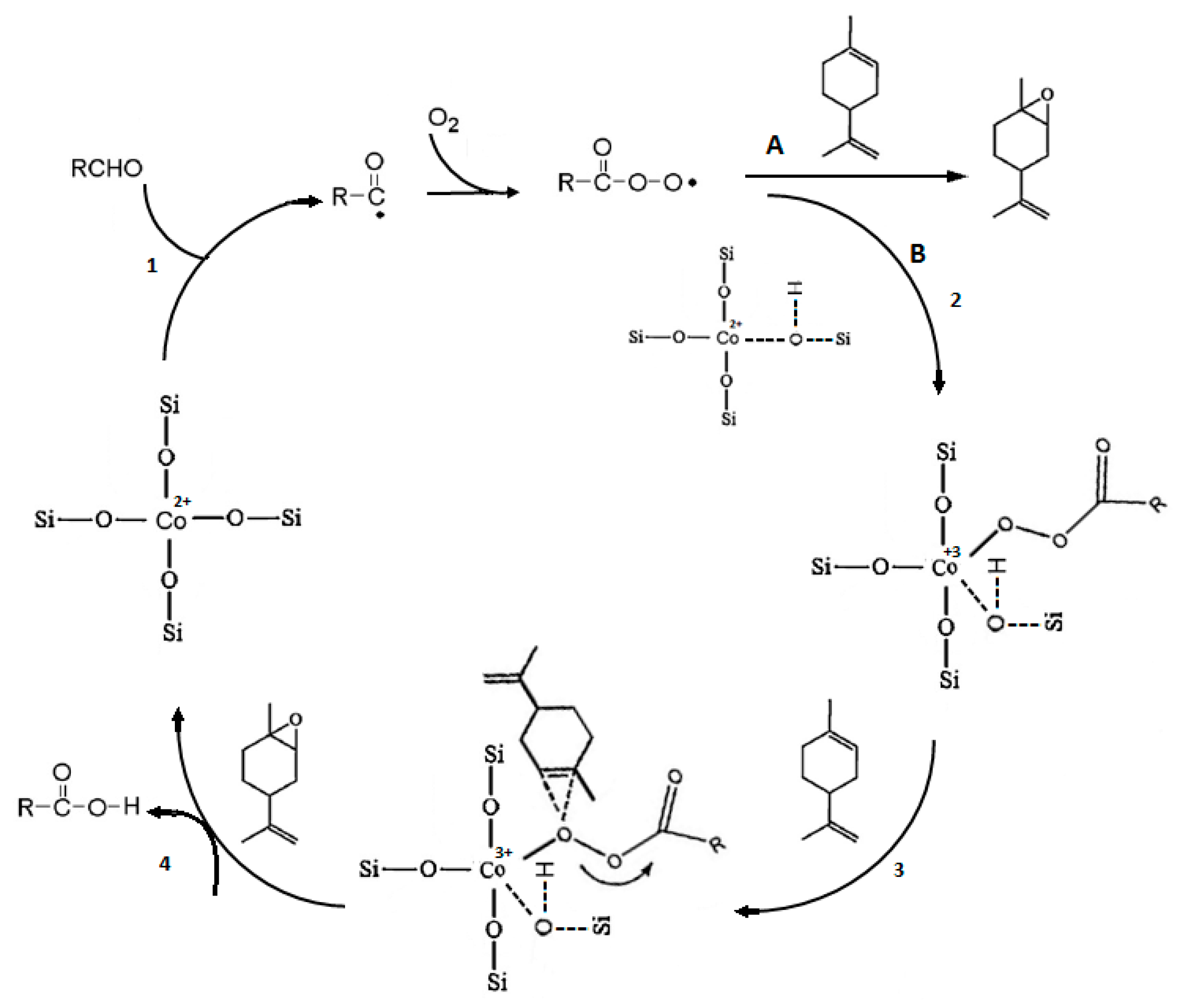
A review of fatty epoxide ring opening reactions: Chemistry, recent advances, and applications - Moser - 2022 - Journal of the American Oil Chemists' Society - Wiley Online Library

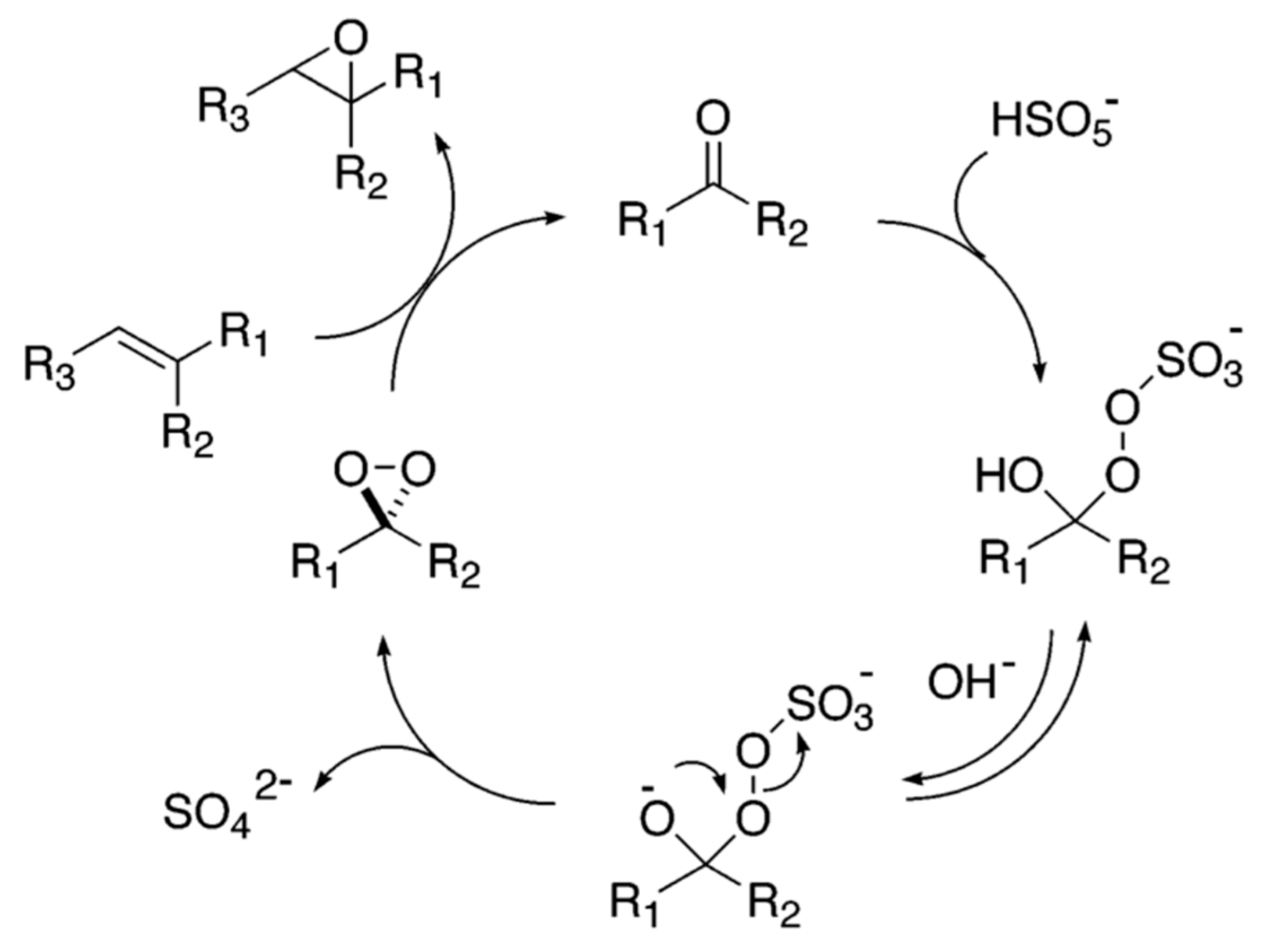
New Insights in the Mechanism of Amine Catalyzed Epoxidation: Dual Role of Protonated Ammonium Salts as Both Phase Transfer Catalysts and Activators of Oxone | Journal of the American Chemical Society

PDF) Development of a Selective, Solvent-free Epoxidation of Limonene Using Hydrogen Peroxide and a Tungsten-based Catalyst

Regioselective Bromination of Organic Substrates by Tetrabutylammonium Bromide Promoted by V2O5−H2O2: An Environmentally Favorable Synthetic Protocol | Organic Letters

EP0434546A1 - Process for the selective epoxidation of unsaturated (meth)acrylic compounds and new obtained bifunctional (meth)acrylates - Google Patents

PDF) Development of rapid and selective epoxidation of α-pinene using single-step addition of H2O2 in an organic solvent-free process | ana lópez fernández - Academia.edu

In situ epoxide generation by dimethyldioxirane oxidation and the use of epichlorohydrin in the flow synthesis of a library of β-amino alcohols | Royal Society Open Science

Highly efficient epoxidation of electron-deficient olefins with tetrabutylammonium peroxydisulfate - ScienceDirect
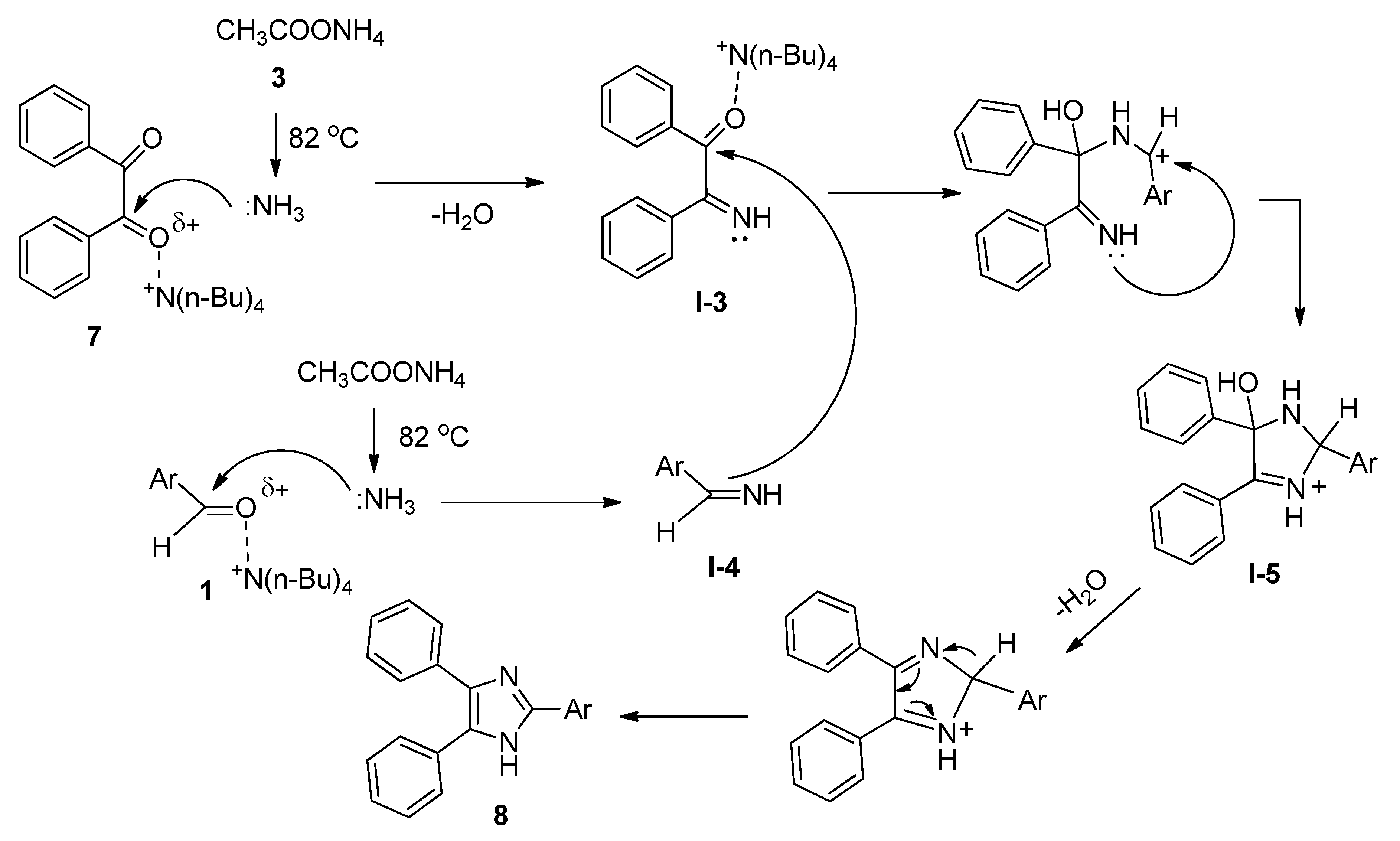
Molecules | Free Full-Text | Tetrabutylammonium Bromide (TBAB) Catalyzed Synthesis of Bioactive Heterocycles

New Insights in the Mechanism of Amine Catalyzed Epoxidation: Dual Role of Protonated Ammonium Salts as Both Phase Transfer Catalysts and Activators of Oxone | Journal of the American Chemical Society
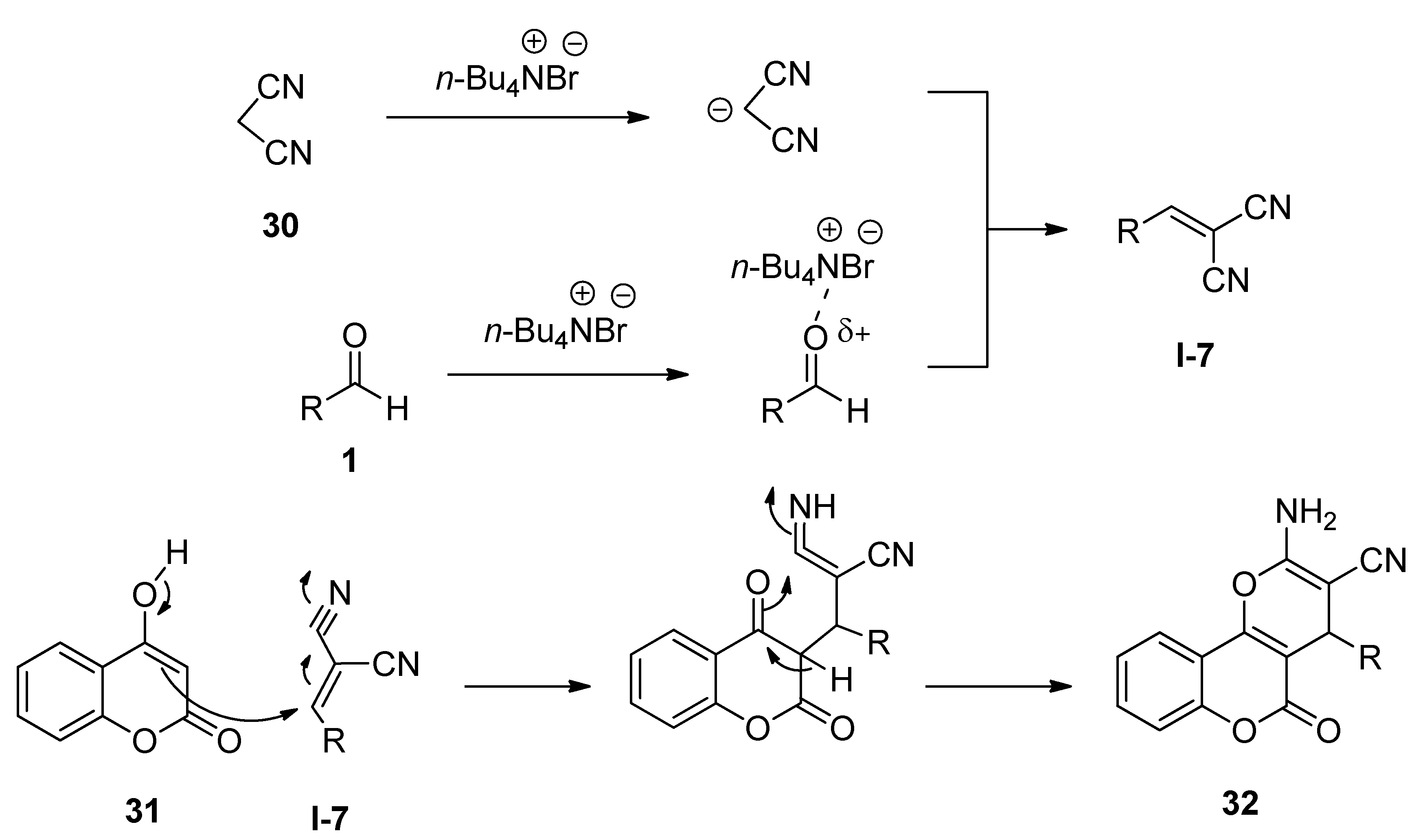
Molecules | Free Full-Text | Tetrabutylammonium Bromide (TBAB) Catalyzed Synthesis of Bioactive Heterocycles

Highly efficient epoxidation of electron-deficient olefins with tetrabutylammonium peroxydisulfate - ScienceDirect