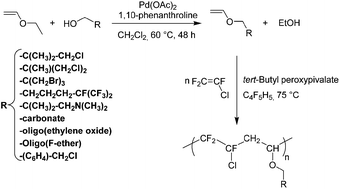
When methyl vinyl ether reacts with a strong acid, the proton adds to C2 exclusively, instead of C1 or the oxygen atom. Draw the three protonated forms of methyl vinyl ether and

2-Chloroethyl vinyl ether, ca. 95%, Thermo Scientific Chemicals, Quantity: 100 g | Fisher Scientific
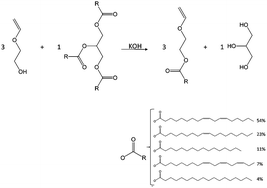
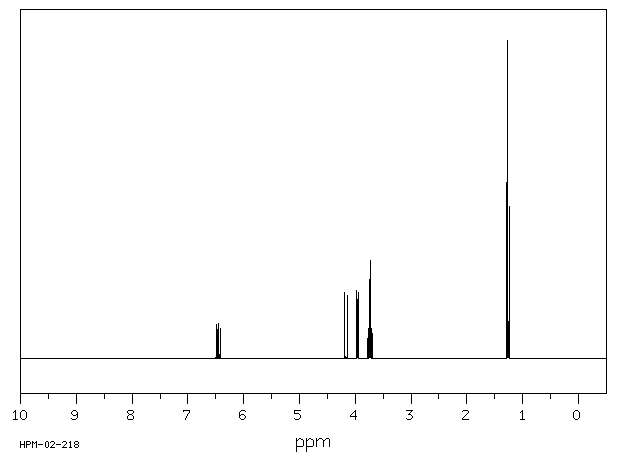
Living carbocationic polymerization of a vinyl ether monomer derived from soybean oil, 2-(vinyloxy)ethyl soyate - Green Chemistry (RSC Publishing)

Expanding vinyl ether monomer repertoire for ring‐expansion cationic polymerization: Various cyclic polymers with tailored pendant groups - Kammiyada - 2017 - Journal of Polymer Science Part A: Polymer Chemistry - Wiley Online Library